By Catherine Jomary, Ph.D., Technology Lead ATMPs, and Cecilia Devlin, Senior Process Architect
Introduction
Since human therapeutics have rapidly evolved from small molecules to include complex biological products such as monoclonal antibodies (mAbs), viral vectors, autologous and allogeneic cell therapies, mRNA therapeutics, drug manufacturers and contract development and manufacturing organizations (CDMO) are considering how to design and operate multiproduct facilities to maintain agility and adaptability. Flexibility, scalability, and speed to market are the key factors driving this endeavor to produce non-viral biologics products, Advanced Therapy Medicinal Products (ATMPs), and mRNA therapeutics. The main challenges are linked to the variability between different technology and manufacturing equipment, diverse processes, logistic requirements, for example, of incoming cells, starting material, and the distribution and delivery of the final products that can differ radically from therapy to therapy. Products targeting rare diseases required usually small batches, but larger batch sizes are required for larger populations affected with common genetic disorders. These different product manufacturing processes are complex, and their specific risks need to be addressed when designing multi-product facilities. Adaptable facilities are required to facilitate rapid change, based on manufacturing needs for different product processes, without significant modification of the facility and quickly add or remove corresponding supporting utility services.
Multiproduct facility constraints
Product manufacturing processes and equipment constraints
Multiproduct facility design needs to consider the key constraints to accommodate a wide range of manufacturing processes, equipment, and biosafety level requirements.
The minimum operational units needed to manufacture these different products can vary from one unit for cell therapy and cell and gene therapy products to three units for mAbs, gene therapy, and mRNAs products (see Figure 1). Each operational unit represents clean room facility space required to perform part of the manufacturing process, either upstream or downstream steps. The room size will be dictated by the equipment size and associated space needed to perform manufacturing operations safely and effectively.
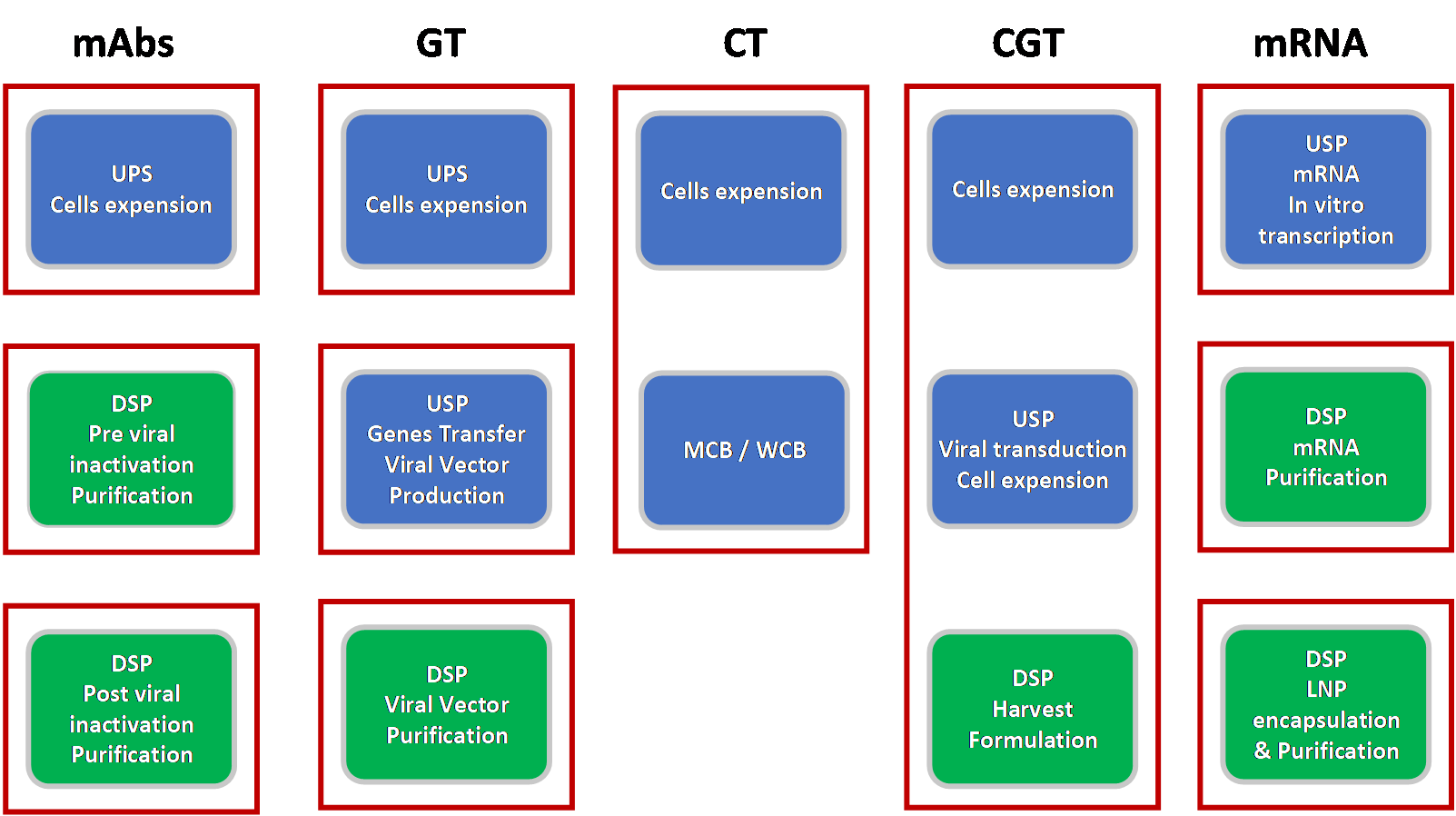
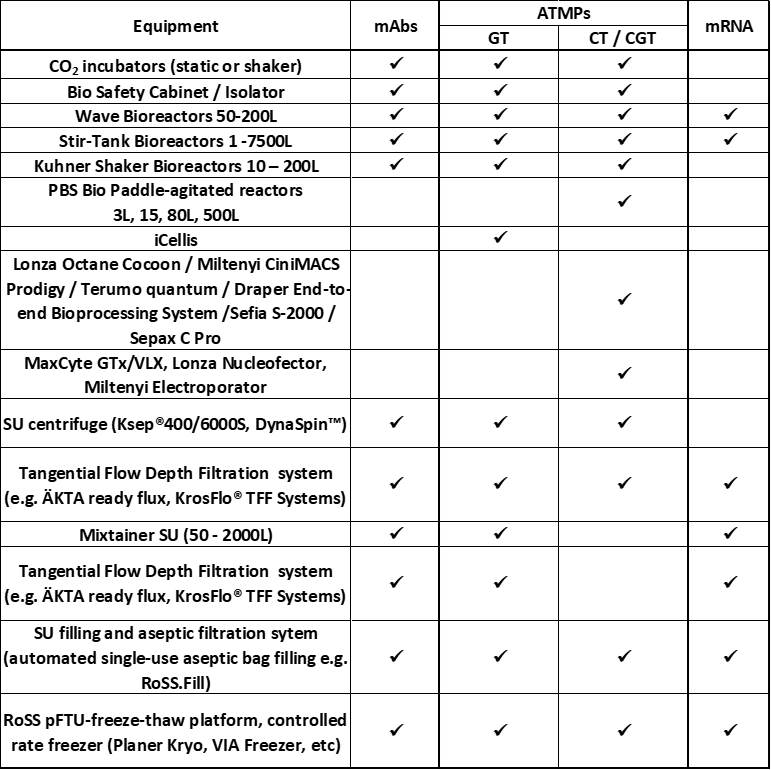
Multiproduct facilities (such as mAbs, ATMPs, and mRNA products) need to be flexible to accommodate various product manufacturing process steps, potential scale-up, and regulatory requirements while maintaining an efficient footprint. It is essential to identify Single Use (SU) equipment that can be utilised for different product processes (see table 1). Evaluation of SU equipment available on the market that can potentially be used for manufacturing these different products showed that a majority of them can be used for mAbs, ATMPs, and mRNA products. Although the size of the equipment is dependent on the product batch size, the same SU equipment can be alternatively used for the manufacture of different products. Some of the equipment utilized for autologous cell therapies that required small-scale batches are specific to this type of product, and consequentially cannot be used for the manufacturing of other ATMPs, mRNAs, and mAbs products.
Operational concept constraints
Operational flexibility within the multiproduct facility must consider all the specific requirements of all product process workflows, including equipment, materials, personnel movements, black utilities, and clean utilities. A key parameter is whether any steps of the manufacturing process are performed open to the environment, briefly exposed, closed, or functionally closed. The selection of single use equipment that permits the closure of the manufacturing process is essential in a multi-product facility. Single-use technology components come pre-sterilized and can help minimize changeover time between products because there is never contact between product and equipment. They do not require clean-in-place activities during the changeover and cleaning validation. The risk of cross-contamination, downtime, and cleaning activities are definitely reduced. Considering that single use equipment, such as bioreactors/reactors, mixtainers, chromatography and TTF systems, can be used alternatively for the manufacturing of mAbs, ATMPs and mRNA products. When portable, they are ready-to-use equipment and can be easily deployed as needed in the different process clean rooms (Table 1).
Since mRNA manufacturing is a cell-free process and highly sensitive to enzymatic degradation, segregation from cell-culture-based ATMPs and mAbs manufacturing is recommended.
Cell-culture inoculation and expansion process steps could potentially be performed in the same containment part of the facility for initiating the manufacturing of ATMPS, mAbs, and therapeutic proteins. However, in the case of gene therapy products, once production of viral vectors is initiated, manufacture is required to be conducted in a spatially segregated area within the multi-product facility to protect the non-viral products from contamination.
If isolators are used for manufacturing viral vectors, they must be decontaminated to inactivate any residual vector presence prior to equipment opening and changeover. Manufacturing of multiple gene therapy viral vectors should be performed either on a temporally segregated campaign basis or in parallel but in completely segregated viral cleanroom areas for other viral products. Unidirectional flows of personnel, materials, products, and waste should be favoured to mitigate the risk of viral contamination being spread throughout the multi-product facility. Contaminated dry and liquid waste needs to be inactivated – chemically or thermally- prior to leaving the containment biosafety level 2 area.
Media and buffers used for the manufacturing of different types of products can be prepared in communal supporting areas ready to be transferred via AT-Port™ System suitable for aseptic transfer directly to all cleanrooms through the ceiling or walls, reducing the cross-contamination risks associated with the flow of buffers and media totes through the clean room corridors.
Extensive cleaning procedures are generally required in the case of manufacturing of viral products, such as airborne peroxide methodology, at change over phase between different products.
Multiproduct Facility Layout Concept
The design of a multiproduct facility must address the operational manufacturing constraints to reduce product-to-product cross-contamination through the separation of product operational units.
A basic cleanroom layout provides a unidirectional flow of personnel and material. Personnel enters a gowning area, from which they can enter a manufacturing-controlled corridor leading to the different cleanrooms. Through a separate airlock (PAL In), personnel enters the operational unit cleanroom to perform manufacturing activities for a specific product. Personnel exit the cleanroom through a separate airlock (PAL Out), and remove their exposed gowning (see figure 2). The different cleanrooms need to be separated by airlocks but do not necessarily be physically separated in the facility. Personnel gowning procedures in place define and control the entry of Personnel to the different manufacturing cleanrooms. Care should be taken to make sure staff will not enter different product cleanrooms without fully de-gowning and re-gowning taking place.
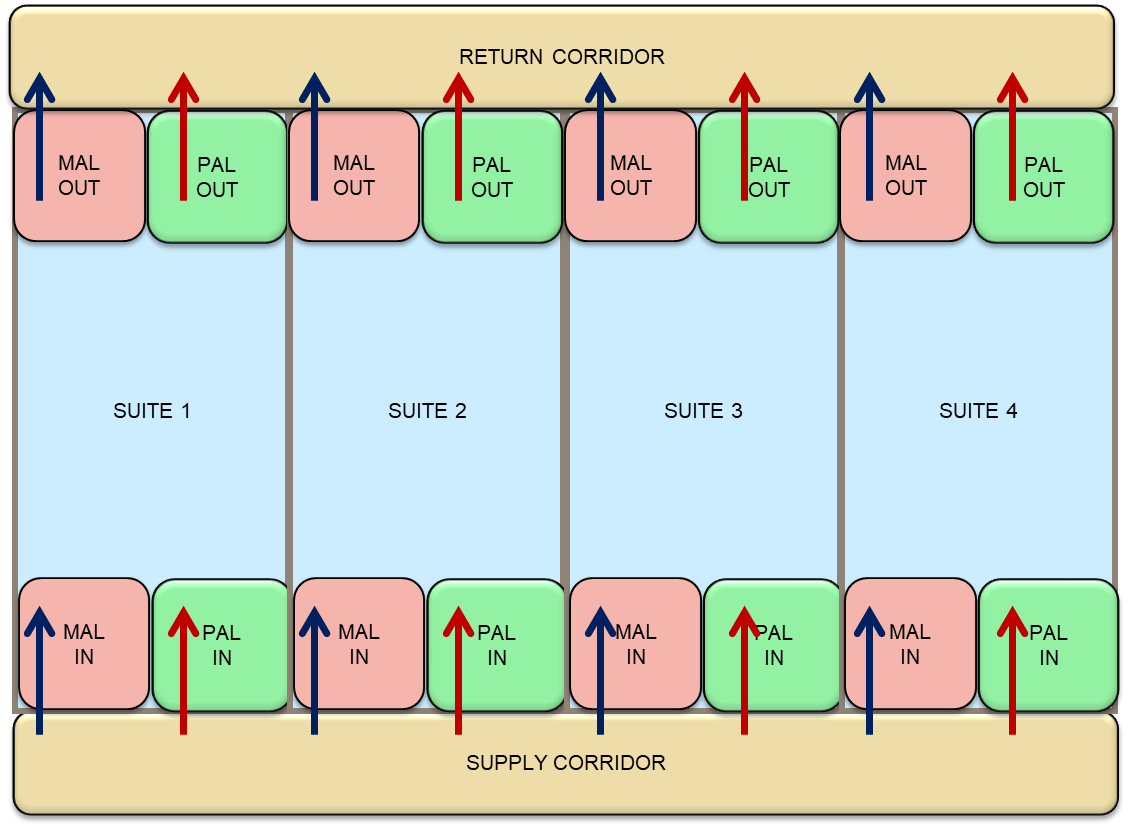
Physical operational unit segregation of both operational units and cleanroom ventilation systems will decrease potential product cross-contamination. An efficient heating, ventilation, and air conditioning (HVAC) system will provide cleanliness, zone segregation/containment, essential for product and personnel safety. The key element is to control the flow of air and prevent aerosolizing of materials and products that can potentially be disseminated through the facility. This can be achieved by implementing a corridor air-handler system that maintains air-pressure cascades. Corridor air pressure always needs to be slightly higher than adjacent rooms and airlocks. Each cleanroom will be equipped with a single-pass system that imports 100% outside air. High-efficiency particulate arrestance (HEPA) filters need to be installed on both incoming and outgoing air to circumvent air duct contamination. In addition, prefilters will have to be changed each time a new product is manufactured in the cleanroom to avoid potential product cross-contamination. A dedicated air handling system for each operational unit combined with airlock systems and a sound pressurization strategy will efficiently control contaminated air from migrating into the adjacent zones housing different product manufacturing activities and ensure personnel and product safety.
To add flexibility, the cleanroom operational units should ideally be organized side by side with a unidirectional flow of personnel and material (Figure 2). If required, the interconnection between those operational units can be done to create more manufacturing space for example, to set up a ballroom cleanroom design.
Alternatively, an operational unit could be divided into smaller units to accommodate the manufacturing of a small batch of products see figure 3 (i.e. to manufacture personalized medicine). To facilitate these spatial changes, multiple connections for clean utilities, media and buffer, and AT ports should be added to the cleanroom ceiling and /or walls. Modification to the cleanroom configuration will have minimal impact on manufacturing operations. A central supply station feeding individual cleanroom operational units with media and buffer from the ceiling will facilitate the changes. In this example, material and personnel flows will become bidirectional.
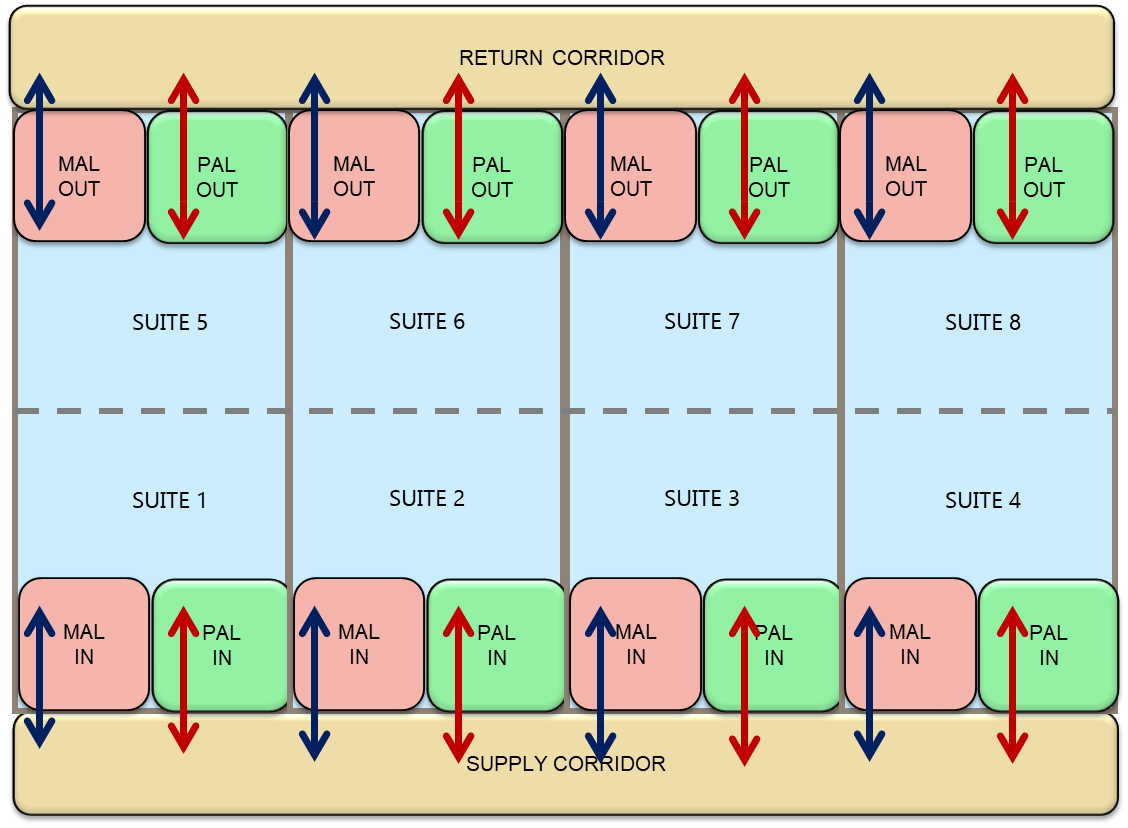
Figure 3: Multiproduct Facility – Bi-directional Flow Double red arrow: flow of personnel- Double blue arrow: flow of material
The cleanroom design approach can be based on a flexible wall panel design that can be reconfigured to reorganize the cleanroom space according to the changes required for the manufacturing of a new product. Therefore, having these cleanroom operational units organized in sets gives the facility added flexibility to accommodate evolving needs over time in adding additional manufacturing space in the same footprint.
To facilitate cleaning room or operational manufacturing units during product change-over, room walls, and ceiling should be smooth, slick, and sloping surfaces without ledges and grooves. Piping should be contained within the walls and can be sealed off when not in use to eliminate the need for cleaning.
The utility system design needs a strong risk assessment to ensure that water, gases, and clean compressed air are distributed without the risk of cross-contamination. A robust control strategy for monitoring and detecting all potential contaminants rapidly will be necessary to balance flexibility and cost-effectiveness. To ensure continuous service, adding utility redundancy in the multiproduct facility design will be cost-effective when increased capacity is needed in the future.
Summary
Multiproduct facilities design needs to be innovative to allow manufacturing flexibility in order to sustain the ever-changing pharmaceutical market and regulatory expectations. These facilities will require more space and creative architectural design that will eliminate the need to build new facilities for each new emerging therapeutics modality and will permit cost-effective manufacturing.
This white paper was originally published in ISPE Pharmaceutical Engineering.



