To understand the modern life sciences manufacturing process, one must understand how the industry has transformed since the beginning of the “Industrial Revolution.”
A Brief History of Industry
Until the end of the 18th century, the concept of industrial manufacturing had not been realized. Goods were produced through manual labor and were often expensive and in short supply. In 1784, steam power was harnessed for the first time to power weaving looms and began what is referred to as the “Industrial Revolution.”
Modern industrialization can be categorized into four periods or iterations:
- Industry 1.0 – 1780s coal and steam power
- Industry 2.0 – 1870s, electrical power, mass production, assembly line
- Industry 3.0 – 1960s, automation, computers, electronics
- Industry 4.0 – 2010s, cyber-physical systems, IIoT
The Early Days of IT/OT Adoption
The modern life sciences manufacturing process is a complex and sophisticated merger of Information Technologies (IT) and traditional Operational Technologies (OT).
IT and OT were first introduced and implemented in the industrial manufacturing process during the late 1960s (Industry 3.0). Both IT and OT can be comprised of hardware and software systems however, historically, OT would interface with physical systems to monitor and control industrial processes. IT would process, transmit, and store business or other product data.
Since their inclusion in the industrial manufacturing process, IT and OT have remained the underlying technologies critical in supporting the manufacturing process. Traditionally these technologies have remained siloed, advancing in parallel but often considered separate.
In the late 1990s technology began to increase exponentially and with this, we began to see the computerization of systems, equipment, and instruments. With the integration of technology into all aspects of our lives, we also began to see the integrations occurring rapidly in the manufacturing process. The IT/OT integrations began to blur the lines of organizational responsibility and the life sciences industry was faced with challenges regarding data and records that were unique and required new regulations and guidance.
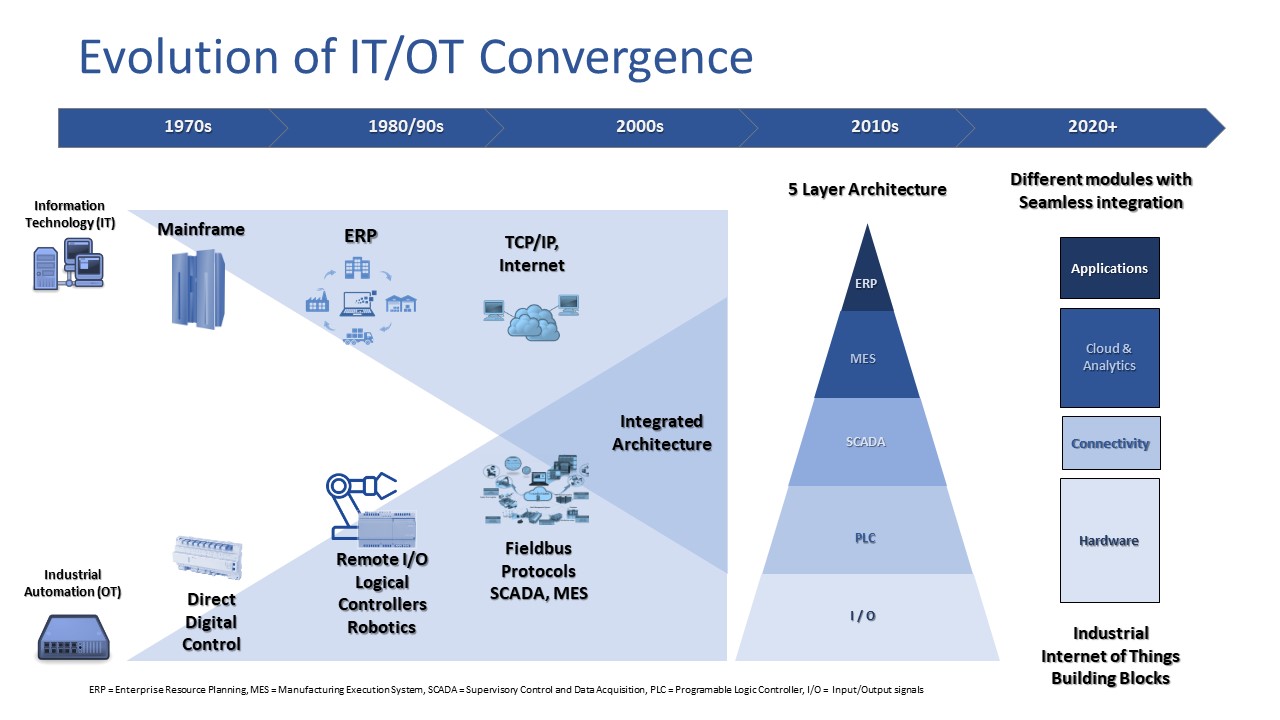
The Modern IT/OT Convergence
As technologies and digital capabilities increased, and IT surpassed OT capacities, the life sciences industry had one foot in the past and one foot in the future. Manufacturers of OT recognized this trend and began integrating IT components into OT to deliver products which enabled their clients increased functionality and reduced costs.
With the increased convergence of IT and OT a different approach to projects often needs to be taken which may result in projects that were driven by engineering may now be owned by the IT department and in some cases, the opposite may be true.
This amalgamation of technologies often requires organizations to rethink many aspects of their operations, specifically around data integrity, infrastructure capacity, networking, and most importantly security: both physical and logical.
Industry 4.0 & Pharma 4.0™
Industry 4.0 is the next iteration of Industry which introduces the cyber-physical and Internet of Things (IoT) technologies in the manufacturing process. Industry 4.0 is the automation of traditional manufacturing and industrial practices utilizing integrated “smart” technologies. Industry 4.0 often implements device-to-device or machine-to-machine communication capabilities, machine learning, artificial intelligence, and other advanced analytics to provide additional dimensions of data and expanded insight into the manufacturing process.
Pharma 4.0™ is the extension of Industry 4.0 principles to the life sciences manufacturing process. While Pharma 4.0™ mirrors Industry 4.0, there are unique challenges that life sciences organizations face due to industry specific regulations and guidance that impact the rate of adoption.
Pharma 4.0™ is predicated on the Industrial Internet of Things (IIoT) where deep data is harnessed from a myriad of connected sensors and devices and used to make instant and automated operational decisions, and to enable business intelligence at a strategic level. Proper implementation of Pharma 4.0™ technologies results in intelligent factories that run smoother, more efficiently and can respond more rapidly to changing market conditions and demands.
Industrial Internet of Things and the Connected Plant
As Pharma 4.0™ is the extension of Industry 4.0, the Industrial Internet of Things (IIoT) is the extension of the Internet of Things (IoT) to the industrial space. IIoT and the connected plant are a concept in which an organization employs smart, connected devices which share, generate, transfer, and potentially store data to remote premise-based systems or cloud (internet) platforms. This data can then be aggregated and analyzed for standard industrial use or potential alternate views of the data.
IIoT often employs standard communication protocols among devices and systems to allow for easier data transfer, resilient connectivity, and redundant failover measures. IIoT may also employ technologies that enable connection to each sensor in a given system or create dynamic production systems.
Pharma 4.0™, IT/OT Convergence, and the Data Dilemma
One of the greatest benefits of deploying Pharma 4.0™ technologies in the manufacturing process is the abundance of new data which can be generated about one’s business and operational processes.
In the life sciences industry, data associated with the production of a product or batch (electronic or paper-based) is considered part of the GxP record. As life sciences organizations begin the modernization of their manufacturing process and implementing new converged IT/OT technologies to eliminate paper-based records, improve efficiency, and gain deeper insights into their operations, the importance of the integrity and management of data becomes more critical.
If one was to consider an Industry 3.0 manufacturing process versus a similar Pharma 4.0™ manufacturing process in life sciences and the data that is generated from PLCs, SCADA, or similar systems would vary drastically between them. The below image depicts the relative number of data tags (I/O inputs) attributed to a Pharma 3.0 manufacturing facility versus a Pharma 4.0™ manufacturing facility.
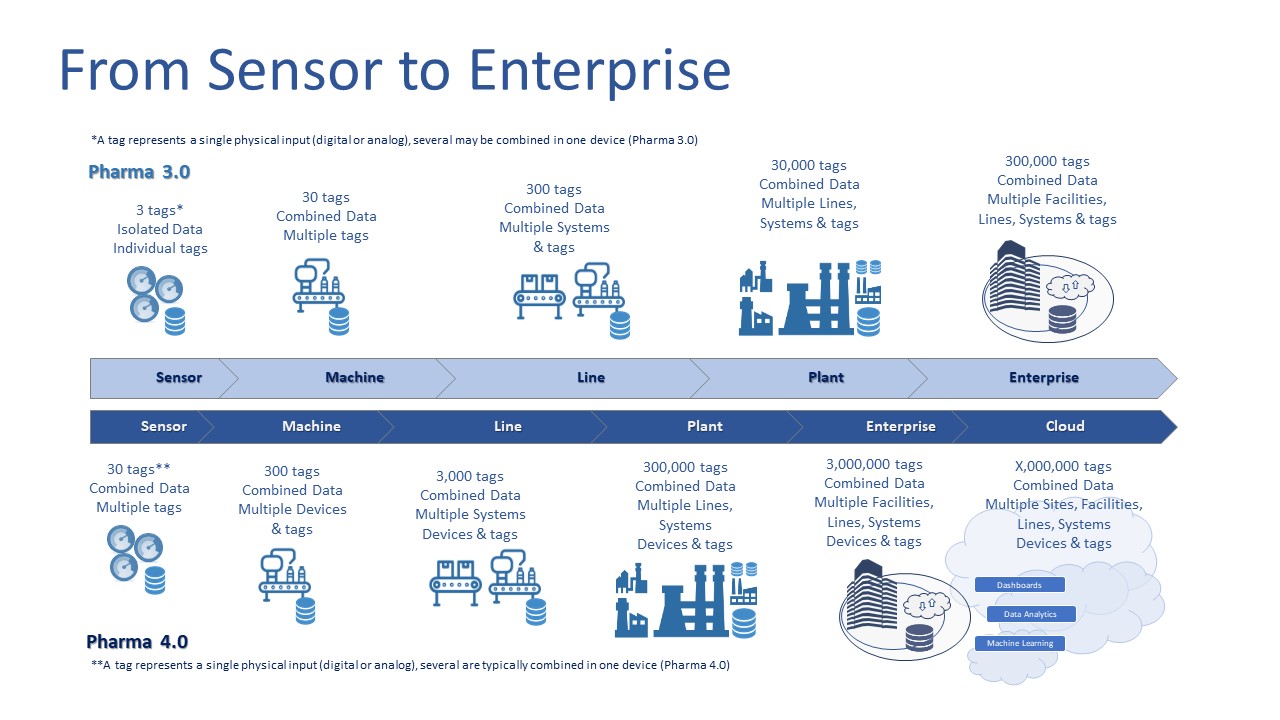
The Pharma 3.0 and 4.0 facilities and processes are similar in many perspectives, but the complexity rises exponentially with an increase in available sensor parameters (data) and integration in Pharma 4.0™ architectures. Pharma 4.0™ also adds the additional layer of on-premise and / or internet/cloud connectivity and data analytics.
The potential increase in the volume and resolution of data generated in the Pharma 4.0™ manufacturing process can be ten times that of the Pharma 3.0 process. This volume and complexity of data is one of the reasons that we see slow adoption by many manufacturers, or full to partial adoption by only larger life sciences organizations with abundant resources.
To fully benefit from the next generation of pharmaceutical manufacturing technologies and practices, a life sciences organization must undergo a digital transformation of its operations which can only be achieved through Digital Maturity and incorporation of Data Integrity principles into all aspects of the operation (Data Integrity by Design). These have been identified as the two key enablers of the ISPE Pharma 4.0™ Special Interest Group’s (SIG) “Holistic Control Strategy.”
Digital Maturity
Digital Maturity is a term that can be difficult to explain or define. To a life sciences manufacturing organization, it is often conveyed as one’s ability to leverage data across the operational and informational technology business platform in a manner that enables transparency, predictability, and adaptability. In plain terms, Digital Maturity speaks to an organizations ability to access, understand, and make key operations and business decisions based upon the available information at hand.
One’s Digital Maturity can be measured and categorized based upon the following criteria:
Level 1: Computerization – The initial and basic level of Digital Maturity, the objective of computerization is to introduce digital technology to automate simple manual processes.
- Achieving level 1 involves finding repetitive tasks that would be better performed by computers and create the basis for a digital infrastructure.
Level 2: Connectivity – Connectivity is focused on streamlining the manufacturing, IT, and business platforms.
- Achieving Level 2 requires manufacturing to expand IT infrastructure and lay the foundation to integrate IT, manufacturing, and business platforms.
Level 3: Visibility – Visibility speaks to the organization’s ability to make decisions based upon comprehensive and complete data being available.
- Achieving Level 3 requires an organization to connect people, machines, and processes to create a substantial digital record of production that can be used to make real-time, data-driven decisions. This is known as cyber-physical systems (CPS).
Level 4: Transparency – At this level of Digital Maturity, the organization is capable of correlating complex interactions between systems, data, and/or processes.
- Achieving Level 4 requires an organization to develop and implement methods to leverage data analytics to interpret the data relationship findings and to understand data impact and effects on the business and operations. With more data resolution, new insights about complex systems become available. Advanced analytics find opportunities for improvement that were previously invisible.
Level 5: Predictability – The second highest level of Digital Maturity, predictability allows the organization to prepare and forecast potential events and avoid potential issues.
- Achieving Level 5 requires that an organization be capable of simulating possible future scenarios through machine learning or AI. Detailed production records enable manufacturers to correct problems before they happen.
Level 6: Adaptability – The highest and most complex level of Data Maturity is adaptability.
- Achieving Level 6 is difficult, often costly and requires the full integration of AI into all business and operational aspects of an organization. This is the final stage of maturity. Here, systems anticipate problems and initiate the proper action by themselves. At their most advanced, these are autonomous self-correcting systems.
For consideration, does adaptability also allow for AI suggested improvements? More than just corrections but also improvements?
Data Integrity by Design
Data Integrity by Design is a desired outcome of digital maturity. Data integrity must be incorporated or considered through each phase of a project or business process. Data integrity should be assessed during the initial planning of a project or business process and should be monitored and addressed during the implementation, operation, and retirement of a system or process that supports the operation.
Data Integrity describes measures used to ensure validity, completeness, and accuracy of a data set (metadata) or all data contained in a system, database, or other constructs. Data integrity assessment is a process, and like many other processes can be explained as a life cycle.
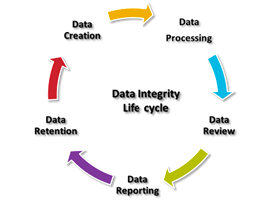
- Data Creation
- Data Processing
- Data Review
- Data Reporting
- Data Retention
When incorporating Data Integrity by Design concepts into one’s project or process, questions should be asked to assess:
Data Creation
- What levels of data to include? Critical Quality Attributes (CQA) and Critical Process Parameters (CPP).
- How does the system generate data?
- Where does/will the data reside?
Data Processing
- Does the data require additional processing or formatting?
- Can the data clearly be utilized for intended use?
Data Review
- How can data be reviewed?
- How often should review be conducted?
- Who will review the data?
Data Reporting
- How will data be represented (on screen/report)?
- If reports are generated, are they accurate and reflect values from the system?
- What format are reports available in and are they indelible?
Data Retention
- How will data be stored?
- How will data be secured?
- Is there a backup and restoration process plan?
- Have regulatory retention considerations for data been addressed?
Planning for the Convergence of the Modern IT/OT
If you consider data as your key output of a Pharma 4.0™ project besides the product you are producing, life sciences organizations should consider several points that may pose potential risks during the planning and implementation phases of a project.
Data Integrity
- As a minimum, the business should perform a data integrity assessment of a system currently in use or that may shortly be implemented. This assessment should be incorporated in the compliance and validation process followed by the business. The assessment is a series of questions relating to:
- data input/output
- data changes/entry
- security roles
- data backup
- critical data fields and audit trail challenges
- electronic signatures
Infrastructure Capacity
- As the backbone of any modern successful organization, the infrastructure which transfers, stores, and archives your data should be assessed for:
- network capacity - ability to handle and manage increased data traffic
- storage capacity - ability to store and manage increased data traffic
- bandwidth capacity - available internet connection speed
- compute capacity - available computing capabilities
Many organizations considering the migration to the new smart technology platforms should invest in infrastructures with a scalable capacity to ensure the ability to upgrade or expand as needed to manage growing volumes of data and communication.
Security (Physical & Logical)
The implementation of Pharma 4.0™ and IIoT technologies begins to make security a bit more challenging from both the physical and logical dimensions. There is an old saying that says, “a chain is only as strong as its weakest link,” the same is true for your network and newly-implemented, always-connected technologies with Pharm 4.0. Ultimately, controlling access to your organization’s data should be your main concern.
Considering that converged IT/OT platforms are interconnected across the business and data is often flowing bidirectionally to and from external third-party providers, securing your data is a much greater concern once it is outside of your control.
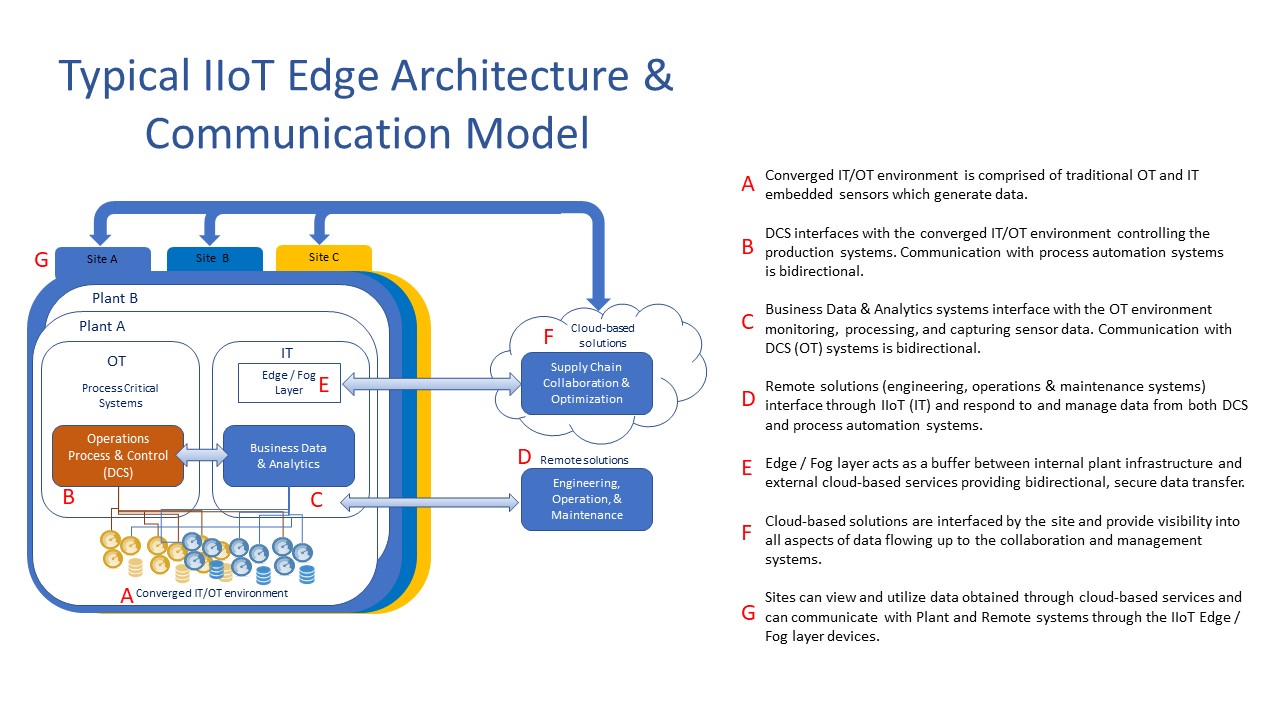
Great caution should be taken to fully understand connectivity and integration points of systems and both technical and procedural controls should be implemented to mitigate any potential risks to your organization. Additional care should be taken when assessing vendors or partners contracted to manage, store, process, or present your data externally to your organization as they may become the system of record for your data in some or all capacities.
Pathway to Digital Maturity (Level 6) and Pharma 4.0™
We have explained the history of industry, IT/OT convergence, Pharma 4.0™, IIoT, data concerns, Digital Maturity, Data Integrity, and additional considerations but now what? What do we do with all of this information and how do we get started?
Step 1 – How mature are we?
The starting point and fundamental requirement to adopting Pharma 4.0™ and IIoT technologies is to determine your organization’s Digital Maturity. Going through the Digital Plant Maturity Model (DPMM) assessment tool1 allows your organization to identify gaps and better understand where resources should be applied.
1Tools exist to assist in this process such as BioPhorum’s Digital Plant Maturity Model: https://www.biophorum.com/resource/digital-plant-maturity-model/downloads/
Step 2 – Organizational Alignment
Organizational alignment is imperative to the successful adoption of Pharma 4.0™ and IIoT technologies. As the end result of a Level 6 digitally mature organization is complex and overarches the entirety of one’s operational organization, support from executive management is critical and recommended as well as obtaining an executive sponsor for the effort.
Step 3 – Assemble Your Tiger Team
Implementing a successful Pharma 4.0™ strategy and rollout requires the “best of the best” within your organization. Identify the key stakeholders within the organization and subject matter experts that will enable your project to succeed. If internal resources are not available, consider third-party assistance from trusted vendors or service providers as they may have experience in implementing and rolling out IT/OT integrated platforms.
Step 4 – Plan, Review, Plan, Review
With any successful project, planning is critical and the implementation of Pharma 4.0™ technologies will test your planning and management skills to their ends. After assembling your team of stakeholders and SMEs draft a plan, develop requirements and divide up tasks for each team member. When the plan is complete, review with the entire team and replan and adjust where needed, then review again.
Step 5 – Bite-sized Chunks
Because of the deep and vast potential implications of Pharma 4.0™ technologies, one should look at the goal of your project and the organizational digital maturity level to determine how to phase your implementation and segregate your project into manageable, realistic goals.
In Closing
Pharma 4.0™ and IIoT are both exciting and challenging concepts to implement for a life sciences organization. There are endless gains to successfully implementing Pharma 4.0™ and IIoT technologies with benefits ranging from reduction of operating and manufacturing costs, improved product quality, increased process efficiency, and many other organization-specific advances that can be had.
Organizations should be careful to plan and identify the proper vendors, suppliers, and resources to assist them in implementing new or improving existing IT/OT hybrid systems to ensure that all challenges can be addressed.
Potential challenges often exist in the following areas:
- Cybersecurity
- Infrastructure scalability (computational limitations, storage, network capacity)
- Regulatory uncertainties
- Resources (costs to implement, personnel technical knowledge)
Organizations may also benefit from the multiple assessment processes which will identify their digital maturity, network and infrastructure capacities, process owners, resources (people, knowledge, budget), and enable the identification of potential gaps in their current operational and business processes.
Following these basic five steps outlined in the previous section and being aware of the potential challenges and risks will not be a guaranteed plan for success but rather it a foundation to ensure the proper awareness and due diligence has been taken to start the process of digital transformation and Pharma 4.0™ implementation.
From Systems to Services, Connect Your Data and Operations
IPS understands the steps to engage the client and vendor in order to design a complete hardware and software solution using IIoT and Cloud technology for our life science clients.
We have experience and expertise in implementing independent and customized solutions, edge computing and cloud-based service, and full integration in the GMP environment.
IPS is a global leader in developing innovative business solutions for the biotechnology and pharmaceutical industries. Through operational expertise and industry-leading knowledge, skill and passion, IPS provides consulting, architecture, engineering, construction management, and compliance services that allow clients to create and manufacture life-impacting products around the world. Headquartered in Blue Bell, PA-USA, IPS is one of the fastest-growing companies servicing the life sciences industry with 1,700 professionals in the US, Canada, Brazil, UK, Ireland, Switzerland, Germany, Singapore, China, and India.



